
The basicity of acetic acid is:
A.1
B.2
C.3
D.4
Answer
233.1k+ views
Hint: We know that the basicity of an acid is the number of replaceable hydrogen atoms that are present in a molecule of that acid. Hence an acid that contains only one replaceable hydrogen atom in its molecule is known as a monobasic acid and its basicity is 1. Similarly, the acid which contains two replaceable hydrogen atoms in its molecule is called a dibasic acid and its basicity is 2. With this concept in mind, we will be solving the given question where we have to first know about the molecular structure of Acetic Acid and then find how many hydrogen atoms it replaces to find its basicity.
Step by step answer: Acetic Acid has a total of 4 hydrogen atoms and 2 carbon atoms with functional group oxygen present in its molecular structure as we can see below. The dissociation formula of acetic acid is also given as follows:
$\text{C}{{\text{H}}_{3}}-\text{COOH }\xrightarrow{{}}\text{ C}{{\text{H}}_{\text{3}}}\text{CO}{{\text{O}}^{-}}\text{ }+\text{ }{{\text{H}}^{+}}$
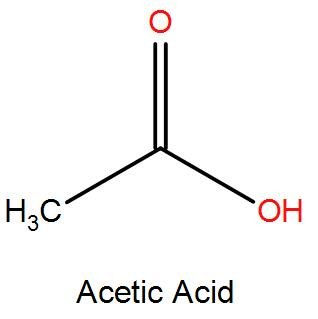
4 hydrogen atoms being present in the molecule of Acetic Acid, it only has one replaceable hydrogen ion present in it. Hence giving only one ionizable ${{\text{H}}^{+}}$ ion, hence its basicity is 1.
So we can determine that the basicity of Acetic acid is 1 because it releases one hydrogen ion in an aqueous solution.
Therefore, the correct option is Option A.
Note: The less electronegative the element, the less stable the lone pair will be and therefore the higher will be the basicity. Even though it’s a weak acid, at high concentrations it will be corrosive on skin. The household vinegar consists of Acetic acid as a primary ingredient.
Step by step answer: Acetic Acid has a total of 4 hydrogen atoms and 2 carbon atoms with functional group oxygen present in its molecular structure as we can see below. The dissociation formula of acetic acid is also given as follows:
$\text{C}{{\text{H}}_{3}}-\text{COOH }\xrightarrow{{}}\text{ C}{{\text{H}}_{\text{3}}}\text{CO}{{\text{O}}^{-}}\text{ }+\text{ }{{\text{H}}^{+}}$

4 hydrogen atoms being present in the molecule of Acetic Acid, it only has one replaceable hydrogen ion present in it. Hence giving only one ionizable ${{\text{H}}^{+}}$ ion, hence its basicity is 1.
So we can determine that the basicity of Acetic acid is 1 because it releases one hydrogen ion in an aqueous solution.
Therefore, the correct option is Option A.
Note: The less electronegative the element, the less stable the lone pair will be and therefore the higher will be the basicity. Even though it’s a weak acid, at high concentrations it will be corrosive on skin. The household vinegar consists of Acetic acid as a primary ingredient.
Recently Updated Pages
JEE Main 2026 Session 2 Registration Open, Exam Dates, Syllabus & Eligibility

JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

Trending doubts
Understanding Average and RMS Value in Electrical Circuits

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

Understanding Atomic Structure for Beginners

Understanding Elastic Collisions in Two Dimensions

For pure water A pH increases while pOH decreases with class 11 chemistry JEE_Main

Which of the following is most stable A Sn2+ B Ge2+ class 11 chemistry JEE_Main

Other Pages
NCERT Solutions For Class 11 Chemistry in Hindi Chapter 8 Redox Reactions (2025-26)

An ideal gas is at pressure P and temperature T in class 11 chemistry JEE_Main

In Carius method of estimation of halogens 015g of class 11 chemistry JEE_Main

Understanding Collisions: Types and Examples for Students

NCERT Solutions For Class 11 Chemistry in Hindi Chapter 1 Some Basic Concepts of Chemistry (2025-26)

Happy New Year Wishes 2026 – 100+ Messages, Quotes, Shayari, Images & Status in All Languages




