
The ratio of sigma and pi bond in mesitylene is:
(A) $ 3 $
(B) $ 7 $
(C) $ 5 $
(D) $ 6 $
Answer
524.7k+ views
Hint :Here, we have to understand the meaning of sigma and pi bonds and then have to draw the structure of mesitylene and then have to count the sigma and pi bonds in the structure then only we can be able to draw the ratio of sigma and pi bonds.
Complete Step By Step Answer:
let us first understand the meaning of sigma and pi bonds as below:
Sigma bond or $ \sigma - $ bond: This is the single bond between two or more atoms with which they are connected by sharing a pair of electrons between them.
Pi-bond or $ \pi - $ bond: There is presence of the double bond between two or more atoms and these bonds satisfy the valence between them out of which one is known as sigma and the other is $ \pi - $ bond.
Now, let us draw the structure of mesitylene compound and it is also known as $ 1,3,5 - {\text{trimethylbenzene}} $ as below:
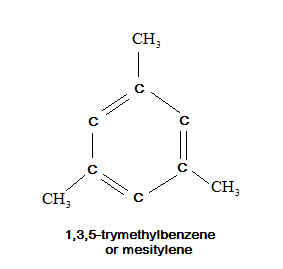
Now, we have to count all the sigma and pi bonds in this structure to calculate the ratio between them but in above structure we see the compactness so we will draw it with more open structure as below:
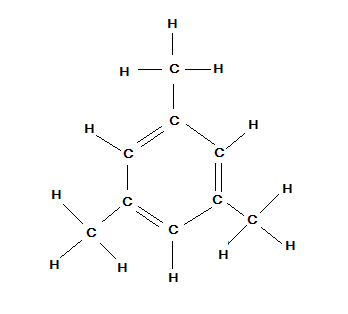
Now, let us count all the single bonds and pi- bonds in the above structure we get:
$ \sigma - bonds = 21 $
$ \pi - bonds = 3 $
Ratio is obtained between them as:
$ \sigma :\pi = 7:1 $ or $ \sigma :\pi = 7 $
Thus, the correct answer is option B.
Note :
Just note the additional information about triple bonds that just like double bonds having one sigma and one Pi bonds the triple bonds also have one sigma and two pi- bonds whereas pi bonds are referred to as the cloud of electrons. To understand this read about pi-bonds and understand the concept.
Complete Step By Step Answer:
let us first understand the meaning of sigma and pi bonds as below:
Sigma bond or $ \sigma - $ bond: This is the single bond between two or more atoms with which they are connected by sharing a pair of electrons between them.
Pi-bond or $ \pi - $ bond: There is presence of the double bond between two or more atoms and these bonds satisfy the valence between them out of which one is known as sigma and the other is $ \pi - $ bond.
Now, let us draw the structure of mesitylene compound and it is also known as $ 1,3,5 - {\text{trimethylbenzene}} $ as below:

Now, we have to count all the sigma and pi bonds in this structure to calculate the ratio between them but in above structure we see the compactness so we will draw it with more open structure as below:

Now, let us count all the single bonds and pi- bonds in the above structure we get:
$ \sigma - bonds = 21 $
$ \pi - bonds = 3 $
Ratio is obtained between them as:
$ \sigma :\pi = 7:1 $ or $ \sigma :\pi = 7 $
Thus, the correct answer is option B.
Note :
Just note the additional information about triple bonds that just like double bonds having one sigma and one Pi bonds the triple bonds also have one sigma and two pi- bonds whereas pi bonds are referred to as the cloud of electrons. To understand this read about pi-bonds and understand the concept.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




