
The stability order of carbocation is:
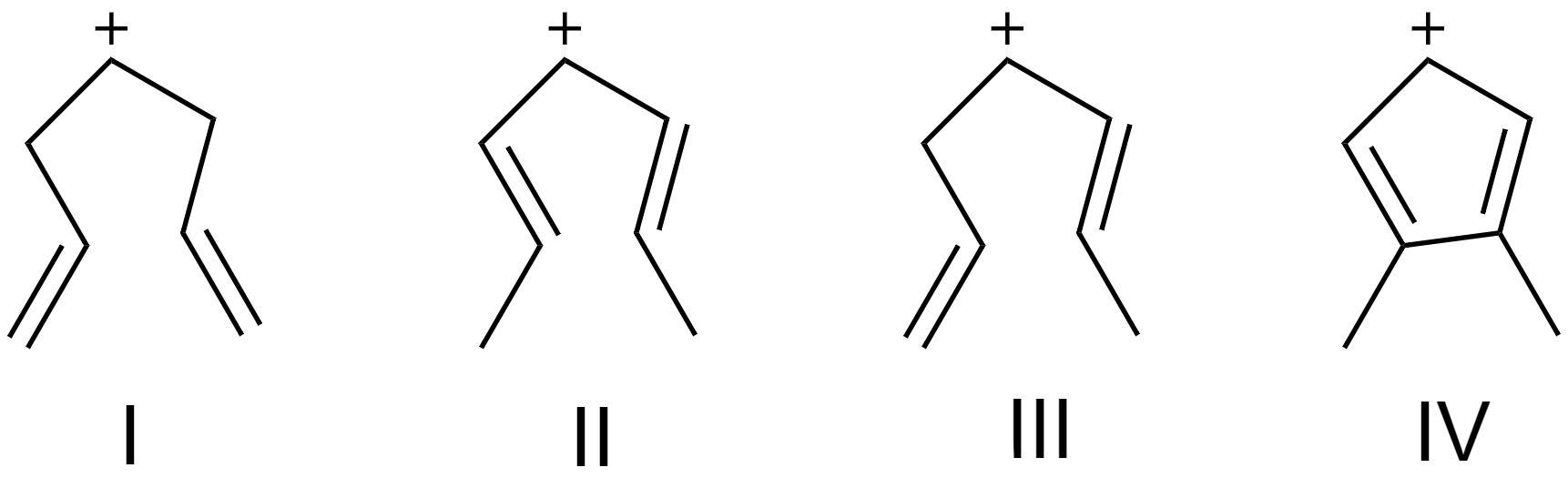
(A) II > IV > III > I
(B) IV > II > III > I
(C) II > III > I > IV
(D) I > III > II > IV

Answer
533.7k+ views
Hint: To determine the stability of these carbocations, we first need to know what carbocations are. A molecule in which the carbon atom forms three bonds and has a positive charge is known as a carbocation. The simplest example of a carbocation is metheniun or methyl carbocation.
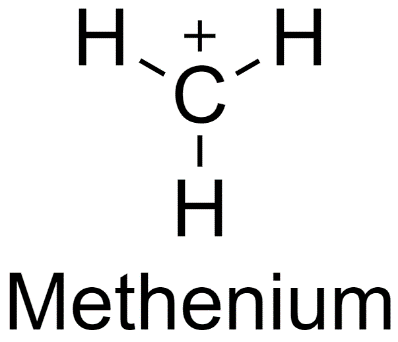
The presence of more than one positive charge on a carbon atom is rare.
Complete answer:
The stability of the carbocation is mainly dependent on three factors
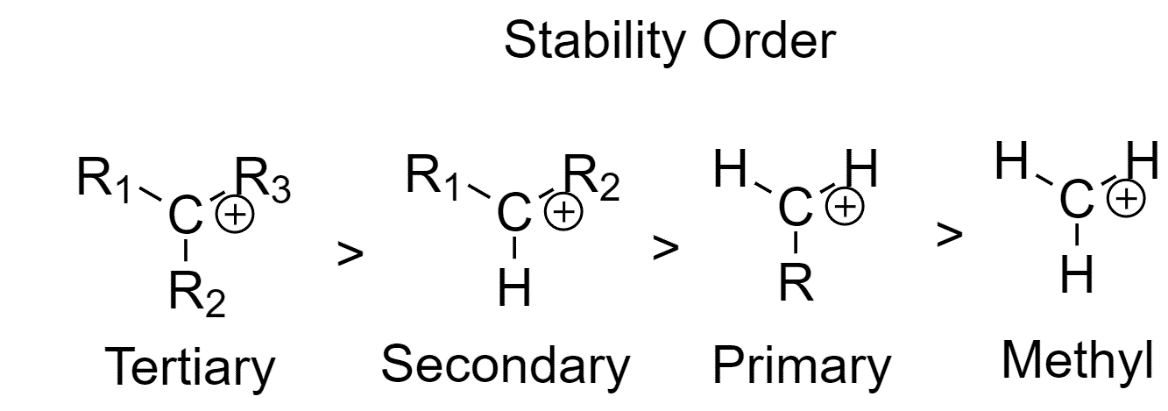
1. The neighboring carbon atoms determine the stability of the carbocation
As we go from primary carbons to secondary carbons and finally to tertiary carbons, the stability of carbocation increases.
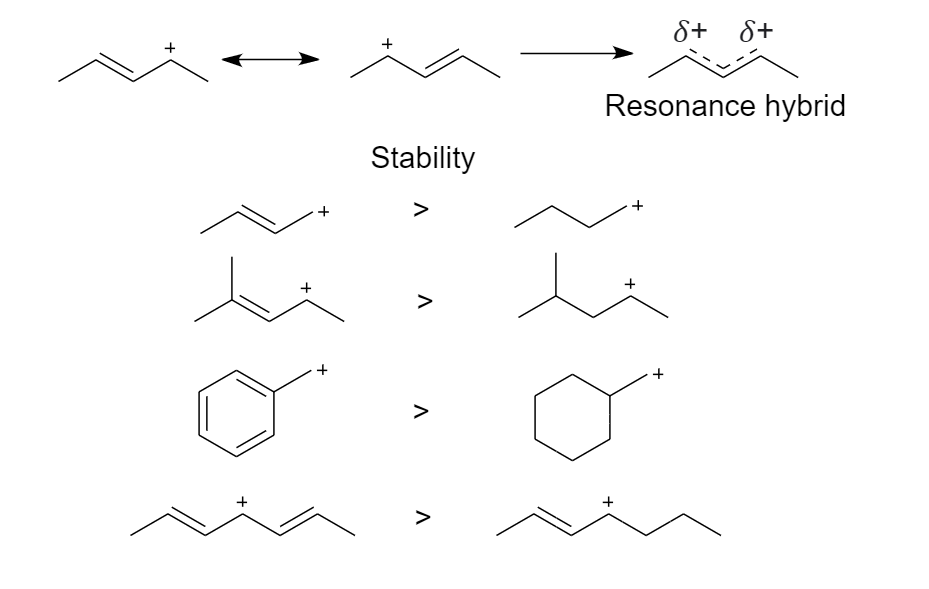
2. The neighboring carbon-carbon multiple bonds stabilize the carbocation
Delocalization occurs when the positive charge is shared between multiple atoms. This happens when the p-orbitals of the $\pi $ bond overlap with the p-orbitals of the carbocation.
Delocalization (resonance) stabilizes a carbocation such that even the most unstable primary carbocation will participate in nucleophilic substitution reactions. It is an additive effect.
3. Adjacent atoms bearing lone pairs stabilize carbocations
When a neighboring atom donates a pair of electrons, $\pi $ donors, there is invariably a formation of a double bond and hence there is an increase in stability of the carbocation.
The basicity of the donor atom affects the stability and hence oxygen and nitrogen are the most powerful $\pi $ donors.
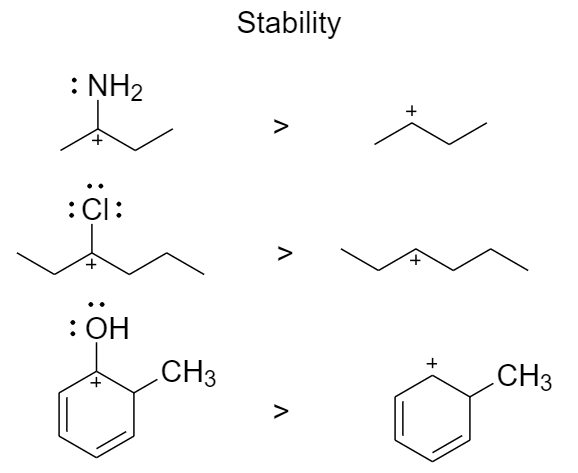
From the above three factors, we can determine that the most stable carbocation is (IV) as it has the most delocalization due to the presence of ring structure.
Followed by carbocation (II) as its two neighboring carbons have double bonds.
Followed by carbocation (III) as one of its neighboring carbon atoms has a double bond.
And the least stable carbocation is (I).
So, the stability order of carbocations is option (B) IV > II > III > I.
Note:
Since carbocation has a positive charge, it only has six electrons in its valence shell and hence is deficient in electrons. It is unstable and is usually formed as an intermediate in substitution reactions, elimination reactions, electrophilic aromatic substitution reactions, etc.
When the stability of a carbocation intermediate is increased, the activation energy of the reaction is lowered, and hence the speed of the reaction increases.

The presence of more than one positive charge on a carbon atom is rare.
Complete answer:
The stability of the carbocation is mainly dependent on three factors
1. The neighboring carbon atoms determine the stability of the carbocation
As we go from primary carbons to secondary carbons and finally to tertiary carbons, the stability of carbocation increases.

2. The neighboring carbon-carbon multiple bonds stabilize the carbocation
Delocalization occurs when the positive charge is shared between multiple atoms. This happens when the p-orbitals of the $\pi $ bond overlap with the p-orbitals of the carbocation.
Delocalization (resonance) stabilizes a carbocation such that even the most unstable primary carbocation will participate in nucleophilic substitution reactions. It is an additive effect.

3. Adjacent atoms bearing lone pairs stabilize carbocations
When a neighboring atom donates a pair of electrons, $\pi $ donors, there is invariably a formation of a double bond and hence there is an increase in stability of the carbocation.
The basicity of the donor atom affects the stability and hence oxygen and nitrogen are the most powerful $\pi $ donors.

From the above three factors, we can determine that the most stable carbocation is (IV) as it has the most delocalization due to the presence of ring structure.
Followed by carbocation (II) as its two neighboring carbons have double bonds.
Followed by carbocation (III) as one of its neighboring carbon atoms has a double bond.
And the least stable carbocation is (I).
So, the stability order of carbocations is option (B) IV > II > III > I.
Note:
Since carbocation has a positive charge, it only has six electrons in its valence shell and hence is deficient in electrons. It is unstable and is usually formed as an intermediate in substitution reactions, elimination reactions, electrophilic aromatic substitution reactions, etc.
When the stability of a carbocation intermediate is increased, the activation energy of the reaction is lowered, and hence the speed of the reaction increases.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

10 examples of friction in our daily life




