
Which would undergo ${\text{S}}{{\text{N}}^2}$ reaction faster in the above pair and why?
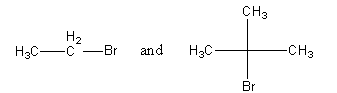
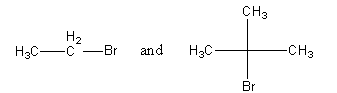
Answer
578.7k+ views
Hint: The ${\text{S}}{{\text{N}}^2}$ reaction removal of a nucleophile and the attack of another nucleophile take place simultaneously. The compound with less steric hindrance will give the ${\text{S}}{{\text{N}}^2}$ reaction faster.
Complete step by step answer:
The full name of ${\text{S}}{{\text{N}}^2}$ reaction is a bimolecular nucleophilic substitution reaction.
In ${\text{S}}{{\text{N}}^2}$ reaction, a nucleophile substitutes another nucleophile.
The whole reaction takes place in one step.
As the nucleophile removes from the reactant another nucleophile attacks from the opposite side. The formed structure is known as a transition state in which both nucleophiles remain bound with weak bonds. Due to the attack from the opposite side the stereo of the product changes. This is known as Walden inversion.
The attacking nucleophile can approach the reactant easily if the reactant has less steric hindrance and thus facilitate the reaction via the ${\text{S}}{{\text{N}}^2}$mechanism.
As the steric hindrance decreases the rate of reaction via ${\text{S}}{{\text{N}}^2}$ mechanism increases.
The order of increasing steric hindrance in alkyl halide is as follows:
${3^ \circ } > \,{2^ \circ }\, > \,{1^ \circ }$
The order of decreasing reactivity of alkyl halide towards the ${\text{S}}{{\text{N}}^2}$ reaction is as follows:
${1^ \circ } > \,{2^ \circ }\, > \,{3^ \circ }$
So, the rate of ${\text{S}}{{\text{N}}^2}$reaction will be high for bromoethane and the rate of ${\text{S}}{{\text{N}}^2}$ reaction will be slow for 2-bromo-2-methylpropane.
Note: The rate of the ${\text{S}}{{\text{N}}^2}$ reaction depends upon both of the reactants. The carbon atom that has a bromide group is attached with only one carbon in bromoethane, so this is known as ${1^ \circ }$ carbon. The 2-bromo-2-methylpropane will favour substitution via ${\text{S}}{{\text{N}}^1}$ mechanism in which a carbocation forms as intermediate. So, the reactivity depends upon the stability of the carbocation. The order of decreasing reactivity of alkyl halide towards the ${\text{S}}{{\text{N}}^1}$ reaction is as follows: ${3^ \circ } > \,{2^ \circ }\, > \,{1^ \circ }$.
Complete step by step answer:
The full name of ${\text{S}}{{\text{N}}^2}$ reaction is a bimolecular nucleophilic substitution reaction.
In ${\text{S}}{{\text{N}}^2}$ reaction, a nucleophile substitutes another nucleophile.
The whole reaction takes place in one step.
As the nucleophile removes from the reactant another nucleophile attacks from the opposite side. The formed structure is known as a transition state in which both nucleophiles remain bound with weak bonds. Due to the attack from the opposite side the stereo of the product changes. This is known as Walden inversion.
The attacking nucleophile can approach the reactant easily if the reactant has less steric hindrance and thus facilitate the reaction via the ${\text{S}}{{\text{N}}^2}$mechanism.
As the steric hindrance decreases the rate of reaction via ${\text{S}}{{\text{N}}^2}$ mechanism increases.
The order of increasing steric hindrance in alkyl halide is as follows:
${3^ \circ } > \,{2^ \circ }\, > \,{1^ \circ }$
The order of decreasing reactivity of alkyl halide towards the ${\text{S}}{{\text{N}}^2}$ reaction is as follows:
${1^ \circ } > \,{2^ \circ }\, > \,{3^ \circ }$
So, the rate of ${\text{S}}{{\text{N}}^2}$reaction will be high for bromoethane and the rate of ${\text{S}}{{\text{N}}^2}$ reaction will be slow for 2-bromo-2-methylpropane.
Note: The rate of the ${\text{S}}{{\text{N}}^2}$ reaction depends upon both of the reactants. The carbon atom that has a bromide group is attached with only one carbon in bromoethane, so this is known as ${1^ \circ }$ carbon. The 2-bromo-2-methylpropane will favour substitution via ${\text{S}}{{\text{N}}^1}$ mechanism in which a carbocation forms as intermediate. So, the reactivity depends upon the stability of the carbocation. The order of decreasing reactivity of alkyl halide towards the ${\text{S}}{{\text{N}}^1}$ reaction is as follows: ${3^ \circ } > \,{2^ \circ }\, > \,{1^ \circ }$.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE

Draw a neat and well labeled diagram of TS of ovary class 12 biology CBSE




