
Write the structure of tetra-tert butyl methane.
Answer
584.7k+ views
Hint: Nomenclature is a very important part of organic chemistry. This nomenclature not only for compounds but also for carbon atoms that make this compound. For nomenclature of carbon atoms, use the terms primary, secondary, tertiary, and quaternary to refer to the substitution level that carbon has in the molecule. Based on the classification of carbon atoms useful to determine the stability and predict the products in organic chemistry reactions.
Complete step by step solution:
Classifications of carbon atoms based on nomenclature are primary, secondary, tertiary, and quaternary. To describe how many carbon atoms are attached to given carbon, this classification is applied to only saturated hydrocarbons and its substitutions compounds like alkyl halides, alcohols, amines, etc.
The terminology of carbon-containing functional groups:
Primary carbon: a hydrogen atom on a carbon attached to one other carbon atom
Secondary carbon atom: hydrogen on a carbon attached to only two other carbon atoms
Tertiary carbon: a carbon atom attached to three other carbon atoms. Giving the name for saturated hydrocarbons starts prefix with tertiary, when a carbon bonded to the other four carbon atoms. This tertiary carbon appears in branched alkanes only, but not in linear alkanes.
Given the structure of tetra-tert butyl methane is:
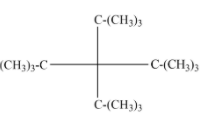
Note: The classification is important as it is applied in the classification of organic compounds with different functional groups. Especially for alcohols, amine, and alkyl halides and also applied for carbocations and carbanions. Due to the hyperconjugation from nearby C-H bonds, tertiary carbocation is more stable compared to primary, secondary, and methyl cation respectively.
Complete step by step solution:
Classifications of carbon atoms based on nomenclature are primary, secondary, tertiary, and quaternary. To describe how many carbon atoms are attached to given carbon, this classification is applied to only saturated hydrocarbons and its substitutions compounds like alkyl halides, alcohols, amines, etc.
The terminology of carbon-containing functional groups:
Primary carbon: a hydrogen atom on a carbon attached to one other carbon atom
Secondary carbon atom: hydrogen on a carbon attached to only two other carbon atoms
Tertiary carbon: a carbon atom attached to three other carbon atoms. Giving the name for saturated hydrocarbons starts prefix with tertiary, when a carbon bonded to the other four carbon atoms. This tertiary carbon appears in branched alkanes only, but not in linear alkanes.
Given the structure of tetra-tert butyl methane is:

Note: The classification is important as it is applied in the classification of organic compounds with different functional groups. Especially for alcohols, amine, and alkyl halides and also applied for carbocations and carbanions. Due to the hyperconjugation from nearby C-H bonds, tertiary carbocation is more stable compared to primary, secondary, and methyl cation respectively.
Recently Updated Pages
Master Class 11 Computer Science: Engaging Questions & Answers for Success

Master Class 11 Business Studies: Engaging Questions & Answers for Success

Master Class 11 Economics: Engaging Questions & Answers for Success

Master Class 11 English: Engaging Questions & Answers for Success

Master Class 11 Maths: Engaging Questions & Answers for Success

Master Class 11 Biology: Engaging Questions & Answers for Success

Trending doubts
One Metric ton is equal to kg A 10000 B 1000 C 100 class 11 physics CBSE

There are 720 permutations of the digits 1 2 3 4 5 class 11 maths CBSE

Discuss the various forms of bacteria class 11 biology CBSE

Draw a diagram of a plant cell and label at least eight class 11 biology CBSE

State the laws of reflection of light

Explain zero factorial class 11 maths CBSE




