
Give conversions: Benzaldehyde to alpha-Hydroxyphenylacetic acid
Answer
539.4k+ views
Hint: Draw the structure of benzaldehyde and $\alpha$-Hydroxyphenylacetic acid and observe the change in conversion of benzaldehyde to $\alpha-$Hydroxyphenylacetic acid. First, benzaldehyde undergoes nucleophilic addition reaction with \[{\text{NaCN}}\] in presence of \[{\text{HCl}}\] and give benzaldehyde cyanohydrins. Acid hydrolysis of cyanohydrins gives rise to alphaHydroxyphenylacetic acid.
Complete answer:
First, we will write the desired reaction.
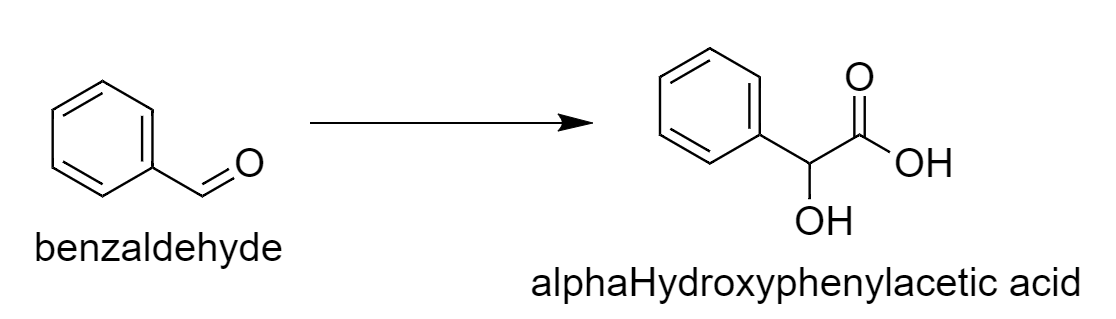
From this reaction, we can say that there is one additional carbon atom of carboxylic acid in the product. So we have to use the reagent that will add one extra carbon atom. So benzaldehyde reacts with \[{\text{NaCN}}\] in presence of \[{\text{HCl}}\] that will increase the carbon atom. The intermediate benzaldehyde cyanohydrins on further reaction with water in presence of acid give rise to alpha-Hydroxyphenylacetic acid.
The detailed mechanism of the reaction is as follows:
Step 1: Nucleophilic addition of \[{\text{NaCN}}\] in presence of \[{\text{HCl}}\] gives Benzaldehyde cyanohydrin.
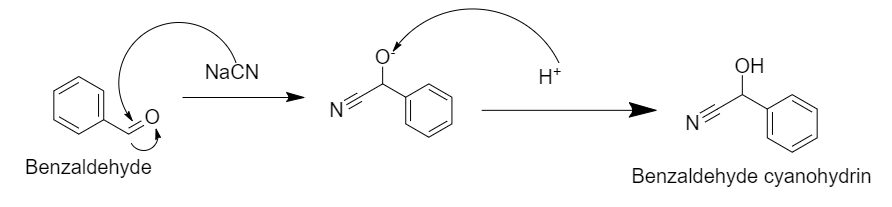
In this reaction, there is a nucleophilic attack of the cyanide group on the carbonyl carbon, which in presence of acid converts into benzaldehyde cyanohydrins.
Step 2: Acid hydrolysis of cyanohydrins gives rise to alpha-Hydroxyphenylacetic acid.
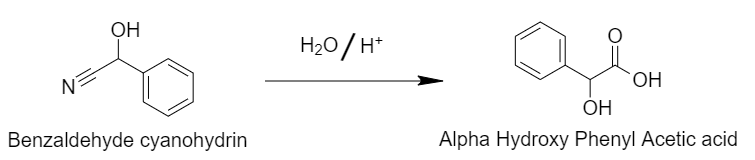
Acid hydrolysis converts the cyanide group of benzaldehyde cyanohydrins to the carboxylic acid group.
Note:
The common name of the alpha-Hydroxyphenylacetic acid is mandelic acid. The IUPAC name of alpha-Hydroxyphenylacetic acid is 2-hydroxy-2-phenyl acetic acid. It has medical applications and is used as an antibacterial agent. Due to the presence of carboxylic and hydroxyl groups, it is a slightly polar compound.
Complete answer:
First, we will write the desired reaction.

From this reaction, we can say that there is one additional carbon atom of carboxylic acid in the product. So we have to use the reagent that will add one extra carbon atom. So benzaldehyde reacts with \[{\text{NaCN}}\] in presence of \[{\text{HCl}}\] that will increase the carbon atom. The intermediate benzaldehyde cyanohydrins on further reaction with water in presence of acid give rise to alpha-Hydroxyphenylacetic acid.
The detailed mechanism of the reaction is as follows:
Step 1: Nucleophilic addition of \[{\text{NaCN}}\] in presence of \[{\text{HCl}}\] gives Benzaldehyde cyanohydrin.

In this reaction, there is a nucleophilic attack of the cyanide group on the carbonyl carbon, which in presence of acid converts into benzaldehyde cyanohydrins.
Step 2: Acid hydrolysis of cyanohydrins gives rise to alpha-Hydroxyphenylacetic acid.

Acid hydrolysis converts the cyanide group of benzaldehyde cyanohydrins to the carboxylic acid group.
Note:
The common name of the alpha-Hydroxyphenylacetic acid is mandelic acid. The IUPAC name of alpha-Hydroxyphenylacetic acid is 2-hydroxy-2-phenyl acetic acid. It has medical applications and is used as an antibacterial agent. Due to the presence of carboxylic and hydroxyl groups, it is a slightly polar compound.
Recently Updated Pages
Master Class 12 Economics: Engaging Questions & Answers for Success

Master Class 12 Physics: Engaging Questions & Answers for Success

Master Class 12 English: Engaging Questions & Answers for Success

Master Class 12 Social Science: Engaging Questions & Answers for Success

Master Class 12 Maths: Engaging Questions & Answers for Success

Master Class 12 Business Studies: Engaging Questions & Answers for Success

Trending doubts
Which are the Top 10 Largest Countries of the World?

What are the major means of transport Explain each class 12 social science CBSE

Draw a labelled sketch of the human eye class 12 physics CBSE

What is a transformer Explain the principle construction class 12 physics CBSE

Why cannot DNA pass through cell membranes class 12 biology CBSE

Differentiate between insitu conservation and exsitu class 12 biology CBSE




