
1, 3-Butadiene with bromine in molar ratio generates predominantly:
(A) 1,1-dibromo-2-butene
(B) 1,4-dibromo-2-butene
(C) 1,2-dibromo-2-butene
(D) 1,3-dibromo-2-butene
Answer
232.8k+ views
Hint: First draw the structure of the molecule and then form the product by keeping in mind the conjugation due to the 2 double bonds present.
Complete step by step answer:
-Let us talk about the structure of 1,3-butadiene. It is a 4 carbon organic molecule with double bonds at 1 and 3 positions. It’s molecular formula is: ${C_4}{H_6}$.
The 2 double bonds make 1,3-butadiene structure to be conjugated (which means there is delocalisation of electrons in this structure). Its conjugated structures are as follows:
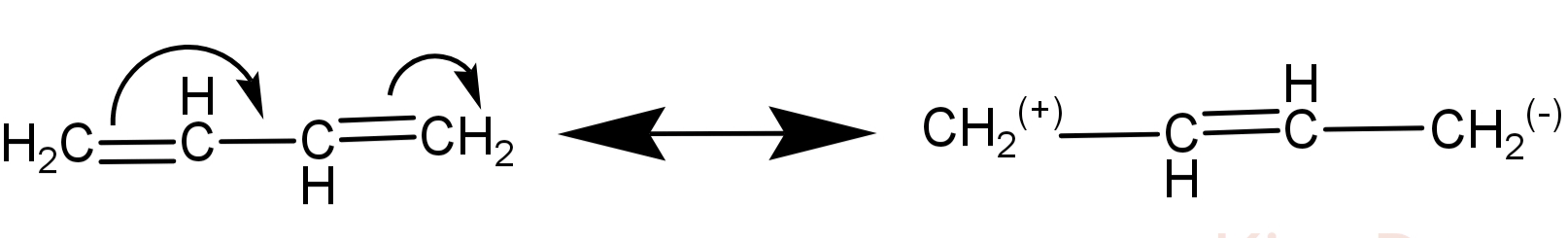
- When halogen attacks on a double bond there is scope for 2 types of products:
One which is formed by 1,2-addition and is kinetically favoured product (because it is formed fast)
And the other is formed by 1,4-addition and is a thermodynamically favoured product because it is associated with thermodynamic stability.
This thermodynamic stability gives a reason for the formation of a 1,4-addition product.
-Also when any halogen attacks on a double bond, there is addition of that halogen on that double bond and a 1,2-addition product might be formed. But this occurs only if there is no conjugation in that double bond.
Here in 1,3-butadiene there is conjugation of double bond and so addition of bromine is 1,4-addition. This means that the double bond shifts between the 2nd and the 3rd carbon (conjugated structure) and Br atoms are added at 1 and 4 position.
The above given reasons favour the formation of a 1,4-addition product. Thus the product formed is 1,4-butadiene.
This is shown in the following reaction:
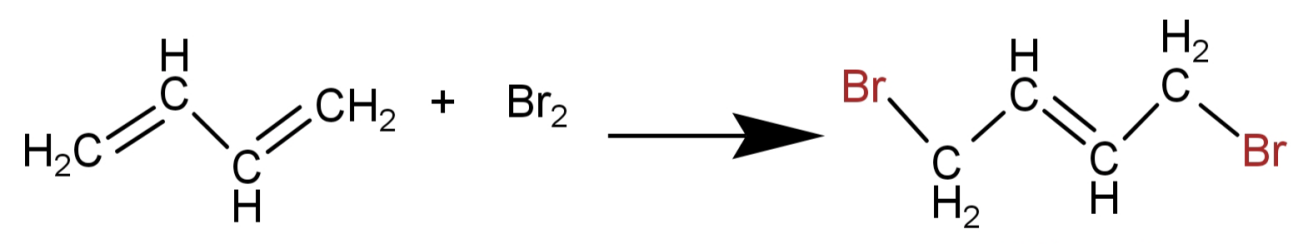
So, the correct option is: (B)1,4-dibromo-2-butene.
Note: Most common mistake we make here is of doing 1,2-addition which means adding 2 Br atoms at 1 and 2 positions of the molecule.
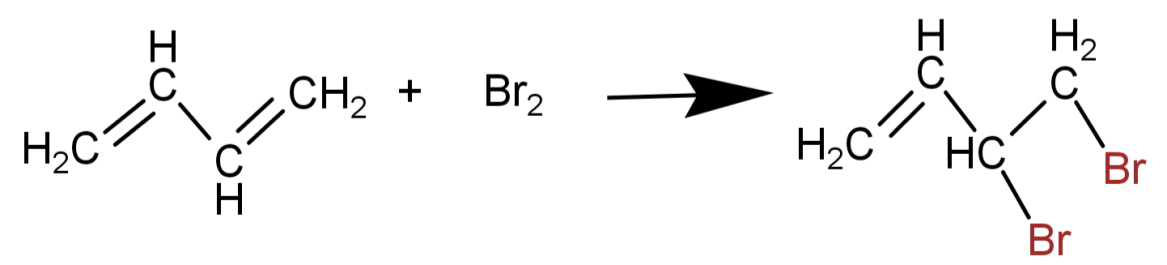
But this is wrong because conjugation is happening here. So, remember that when there is conjugation due to a double bond there will be 1,4- addition.
Complete step by step answer:
-Let us talk about the structure of 1,3-butadiene. It is a 4 carbon organic molecule with double bonds at 1 and 3 positions. It’s molecular formula is: ${C_4}{H_6}$.
The 2 double bonds make 1,3-butadiene structure to be conjugated (which means there is delocalisation of electrons in this structure). Its conjugated structures are as follows:

- When halogen attacks on a double bond there is scope for 2 types of products:
One which is formed by 1,2-addition and is kinetically favoured product (because it is formed fast)
And the other is formed by 1,4-addition and is a thermodynamically favoured product because it is associated with thermodynamic stability.
This thermodynamic stability gives a reason for the formation of a 1,4-addition product.
-Also when any halogen attacks on a double bond, there is addition of that halogen on that double bond and a 1,2-addition product might be formed. But this occurs only if there is no conjugation in that double bond.
Here in 1,3-butadiene there is conjugation of double bond and so addition of bromine is 1,4-addition. This means that the double bond shifts between the 2nd and the 3rd carbon (conjugated structure) and Br atoms are added at 1 and 4 position.
The above given reasons favour the formation of a 1,4-addition product. Thus the product formed is 1,4-butadiene.
This is shown in the following reaction:

So, the correct option is: (B)1,4-dibromo-2-butene.
Note: Most common mistake we make here is of doing 1,2-addition which means adding 2 Br atoms at 1 and 2 positions of the molecule.

But this is wrong because conjugation is happening here. So, remember that when there is conjugation due to a double bond there will be 1,4- addition.
Recently Updated Pages
JEE Main 2023 April 6 Shift 1 Question Paper with Answer Key

JEE Main 2023 April 6 Shift 2 Question Paper with Answer Key

JEE Main 2023 (January 31 Evening Shift) Question Paper with Solutions [PDF]

JEE Main 2023 January 30 Shift 2 Question Paper with Answer Key

JEE Main 2023 January 25 Shift 1 Question Paper with Answer Key

JEE Main 2023 January 24 Shift 2 Question Paper with Answer Key

Trending doubts
JEE Main 2026: Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Angle of Deviation in a Prism

Hybridisation in Chemistry – Concept, Types & Applications

How to Convert a Galvanometer into an Ammeter or Voltmeter

Understanding the Electric Field of a Uniformly Charged Ring

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

Hydrocarbons Class 11 Chemistry Chapter 9 CBSE Notes - 2025-26

Thermodynamics Class 11 Chemistry Chapter 5 CBSE Notes - 2025-26

Equilibrium Class 11 Chemistry Chapter 6 CBSE Notes - 2025-26

Organic Chemistry Some Basic Principles And Techniques Class 11 Chemistry Chapter 8 CBSE Notes - 2025-26

NCERT Solutions For Class 11 Chemistry Chapter 7 Redox Reactions (2025-26)




